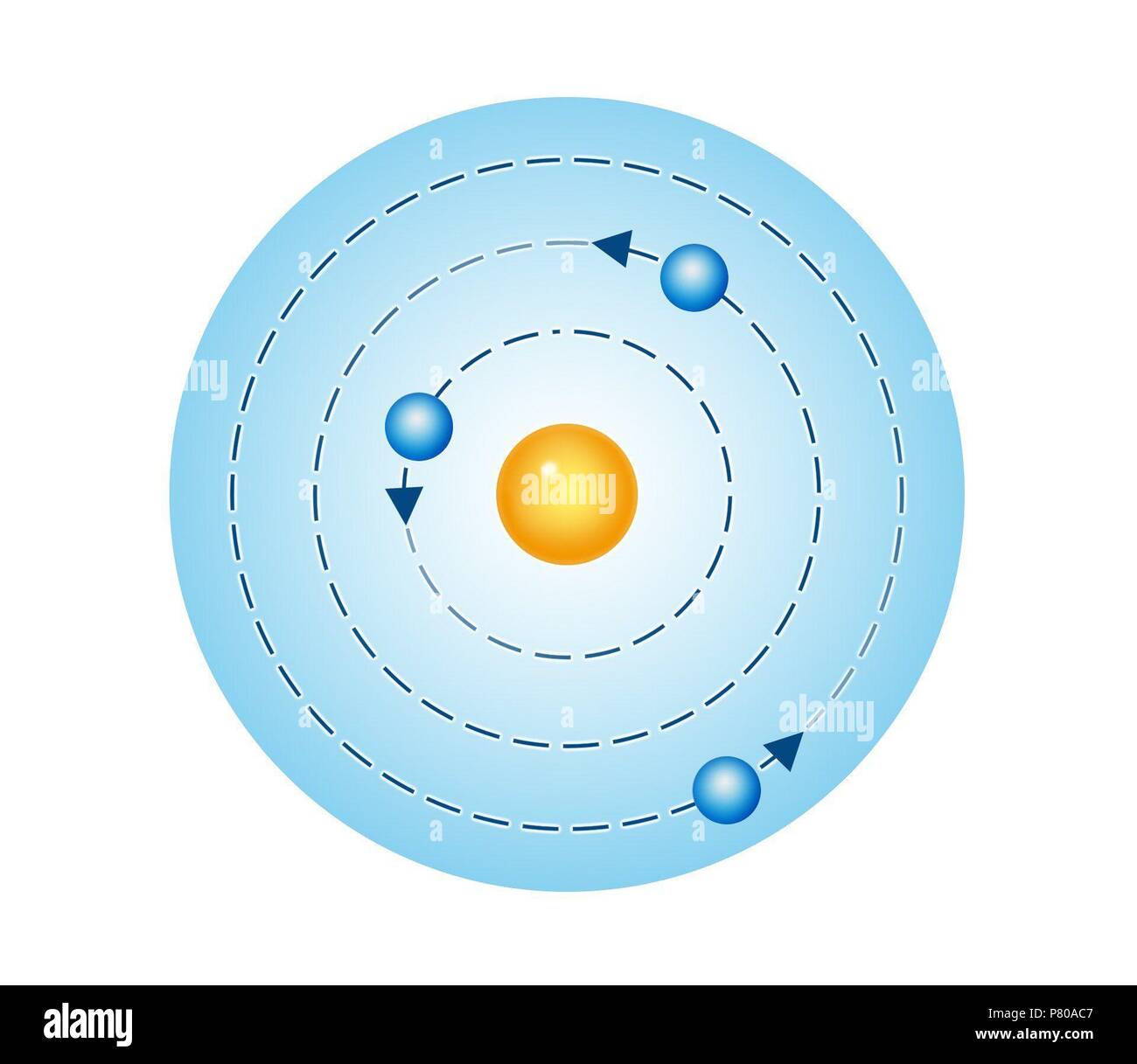
However, the model assumed electrons in the shells didn't interact with each other and couldn't explain why electrons seemed to stack in an irregular manner. It also explained why the noble gases were inert and why atoms on the left side of the periodic table attract electrons, while those on the right side lose them. For example, the shell model explained why atoms got smaller moving across a period (row) of the periodic table, even though they had more protons and electrons. Niel Bohr’s Atomic Theory states that an atom is like a planetary model where electrons were situated in discretely energized orbits. In 1919, Sommerfeld reviewed these achievements in Atombau und Spektrallinien, a book that was soon regarded as the bible of atomic physics. The model explained some of the atomic properties of heavier atoms, which had never been reproduced before. Within a few years of the publication of Bohr’s model in 1913, and despite World War I, atomic and quantum theory was transformed into a budding research field. Bohr began his work on the Manhattan Project after fleeing to Sweden from Denmark because of German occupation in 1943. Thus, the Bohr model for heavier atoms described electron shells. Niels Bohr (1885-1962) was a Danish physicist and winner of the 1922 Nobel Prize in Physics. Once the level was full, additional electrons would be bumped up to the next level. Bohr believed each electron orbit could only hold a set number of electrons. More electrons were required to cancel out the positive charge of all of these protons. One of his best-known students was Ernest Rutherford, who succeeded Thomson as Cavendish Professor of Physics.Heavier atoms contain more protons in the nucleus than the hydrogen atom. Seven of his research assistants, as well as his own son, went on to win the Nobel Prize in Physics. Thomson's greatest contribution to science to be his role as a teacher. Niels Bohr used information from Ernest Rutherfords description of the nucleus of the atom and Max Plancks theory of quantum theory. Thomson published an important monograph in 1913 urging the use of the mass spectrograph in chemical analysis. It seems that he was a wealthy citizen of Abdera, in Thrace that he traveled widely in the. Knowledge of Democritus’s life is largely limited to untrustworthy tradition. His atomic theory helped explain atomic bonding and the structure of molecules. 370) ancient Greek philosopher, a central figure in the development of philosophical atomism and of the atomic theory of the universe. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
